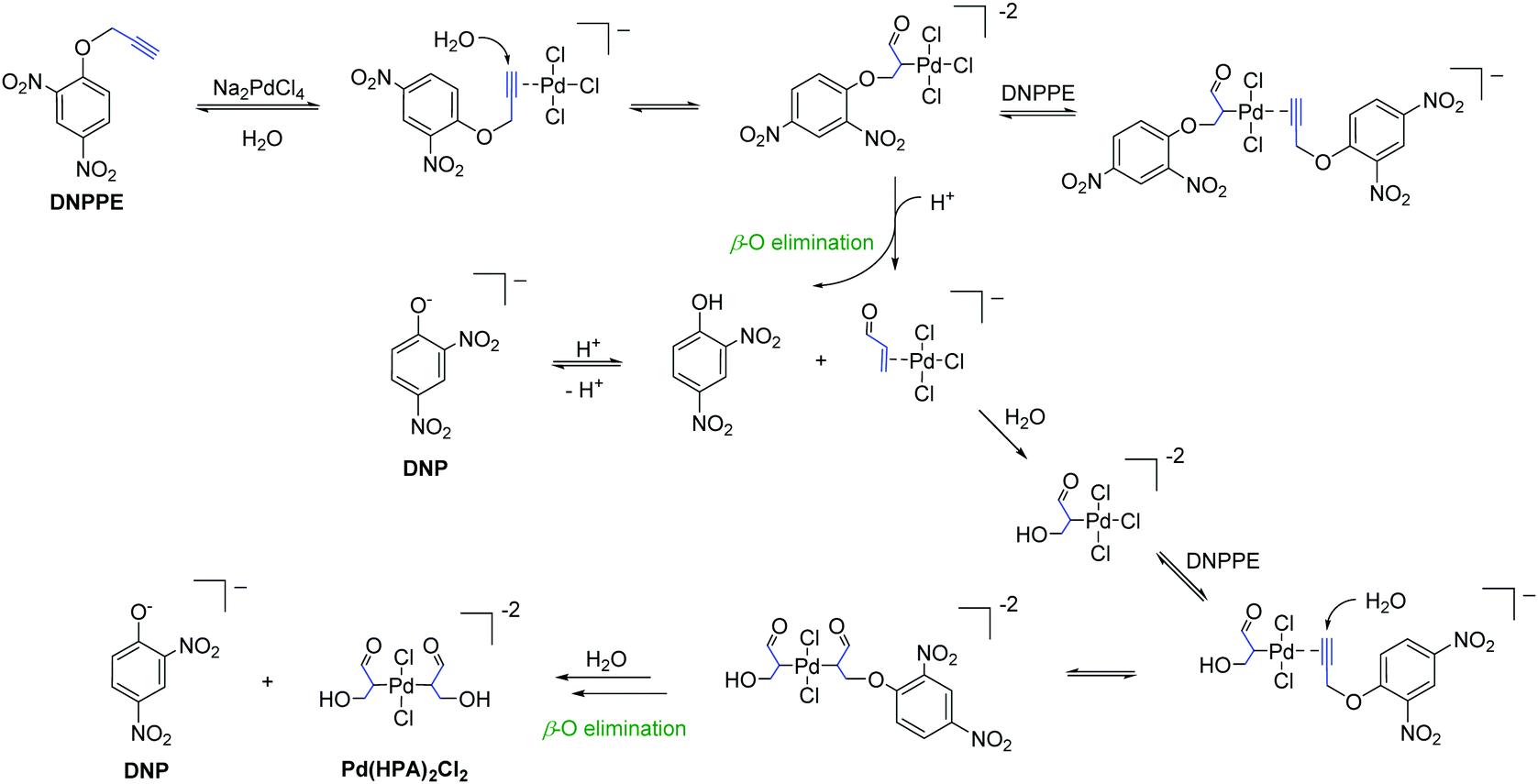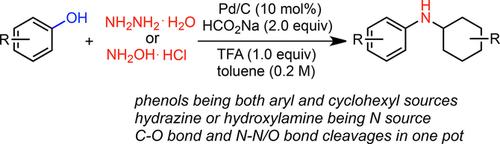
Palladium-Catalyzed Synthesis of N-Cyclohexyl Anilines from Phenols with Hydrazine or Hydroxylamine via N-N/O Cleavage - Adv. Synth. Catal. - X-MOL

Palladium‐Catalyzed Asymmetric Amination of Allenyl Phosphates: Enantioselective Synthesis of Allenes with an Additional Unsaturated Unit - Li - 2014 - Angewandte Chemie International Edition - Wiley Online Library
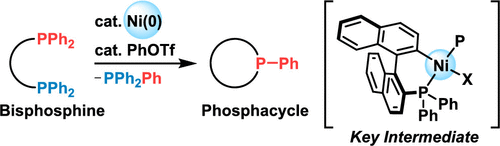
Cyclization of Bisphosphines to Phosphacycles via the Cleavage of Two Carbon–Phosphorus Bonds by Nickel Catalysis,Organic Letters - X-MOL

Enantioselective Decarboxylative Amination: Synthesis of Axially Chiral Allenyl Amines - Wan - 2013 - Angewandte Chemie International Edition - Wiley Online Library
![Palladium‐Catalyzed Cascade C−O Cleavage and C−H Alkenylation of Phosphinyl Allenes: An Expeditious Approach to 3‐Alkenyl Benzo[b]phosphole Oxides - Liu - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library Palladium‐Catalyzed Cascade C−O Cleavage and C−H Alkenylation of Phosphinyl Allenes: An Expeditious Approach to 3‐Alkenyl Benzo[b]phosphole Oxides - Liu - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/b48782c2-6140-4825-86be-d0da17165f14/adsc201800103-toc-0001-m.jpg)
Palladium‐Catalyzed Cascade C−O Cleavage and C−H Alkenylation of Phosphinyl Allenes: An Expeditious Approach to 3‐Alkenyl Benzo[b]phosphole Oxides - Liu - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library
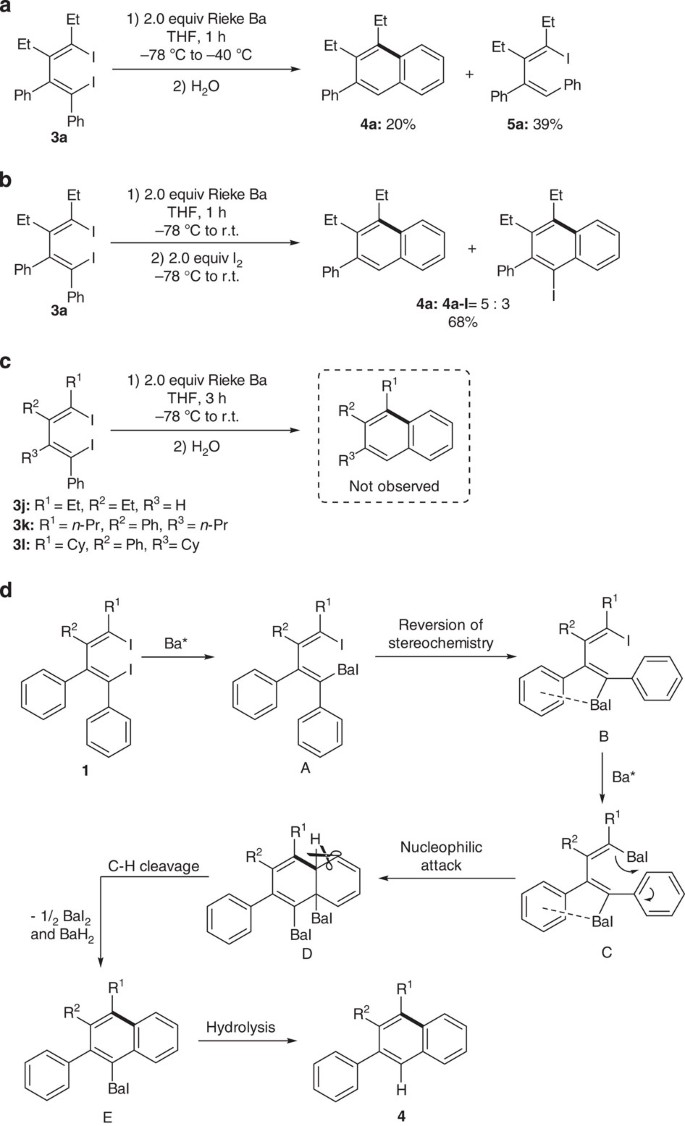
Intramolecular C–F and C–H bond cleavage promoted by butadienyl heavy Grignard reagents | Nature Communications

Palladium: Organometallic Chemistry - William Suggs - - Major Reference Works - Wiley Online Library

Three Carbons for Complexity! Recent Developments of Palladium‐Catalyzed Reactions of Allenes - Lechel - 2013 - ChemCatChem - Wiley Online Library
Unusual C–O bond cleavage of aromatic ethers in ruthenium complexes bearing a 2-alkoxypyridyl fragment† - Dalton Trans. - X-MOL

Facile and selective cleavage of allyl ethers, amines and esters using polymethylhydrosiloxane-ZnCl2/Pd(PPh3)4
Propargylic C(sp3)–H Bond Activation for Preparing η3-Propargyl/Allenyl Complexes of Yttrium,Organometallics - X-MOL

Palladium-Catalyzed Cleavage of α-Allenylic Aryl Ether toward Pyrazolemethylene-Substituted Phosphinyl Allenes and Their Transformations via Alkenyl C–P(O) Cleavage - Org. Lett. - X-MOL
Palladium-Catalyzed Cleavage of α-Allenylic Aryl Ether toward Pyrazolemethylene-Substituted Phosphinyl Allenes and Their Transf

Facile and selective cleavage of allyl ethers, amines and esters using polymethylhydrosiloxane-ZnCl2/Pd(PPh3)4

Molecules | Free Full-Text | The Heck Reaction Applied to 1,3- and 1,2-Unsaturated Derivatives, a Way towards Molecular Complexity | HTML
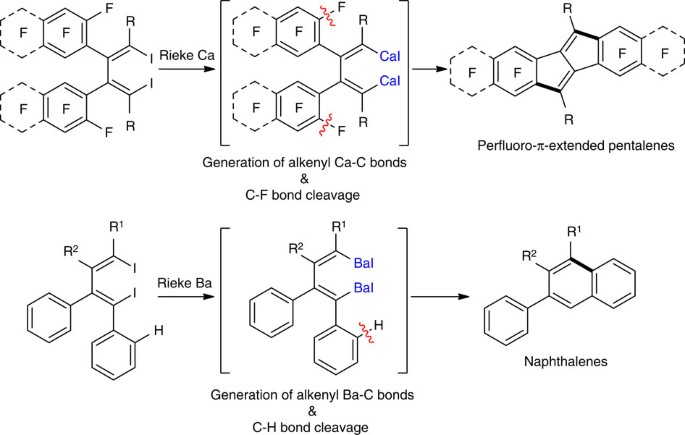
Intramolecular C–F and C–H bond cleavage promoted by butadienyl heavy Grignard reagents | Nature Communications