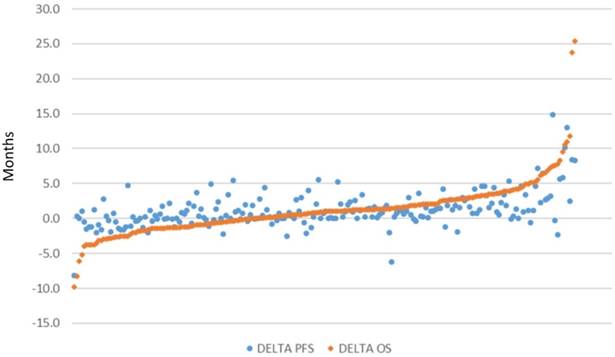
Relationship between Progression-free Survival and Overall Survival in Randomized Clinical Trials of Targeted and Biologic Agents in Oncology

Cancers | Free Full-Text | Prognostic Clinical and Biologic Features for Overall Survival after Relapse in Childhood Medulloblastoma | HTML

Acute myeloid leukemia: survival analysis of patients at a university hospital of Paraná | Hematology, Transfusion and Cell Therapy

A correlation analysis to assess event-free survival as a trial-level surrogate for overall survival in early breast cancer - eClinicalMedicine

OP0294 SJÖGREN'S SYNDROME ASSOCIATED LYMPHOMAS: CLINICAL DESCRIPTION AND 10-YEAR SURVIVAL | Annals of the Rheumatic Diseases

Event-free survival (EFS) and overall survival (OS) according to risk... | Download Scientific Diagram

Wilms tumour event-free and overall survival in Southern and Eastern Europe: Pooled analyses of clinical data from four childhood cancer registries (1999–2017) - European Journal of Cancer

Disease-free survival as a surrogate for overall survival in patients with HER2-positive, early breast cancer in trials of adjuvant trastuzumab for up to 1 year: a systematic review and meta-analysis - The

Relationship between presarcopenia and event occurrence in patients with primary hepatocellular carcinoma | Scientific Reports
Progression-free survival (PFS), event-free survival (EFS), and overall... | Download Scientific Diagram

Event-free and overall survival following neoadjuvant weekly paclitaxel and dose-dense AC +/- carboplatin and/or bevacizumab in triple-negative breast. - ppt download
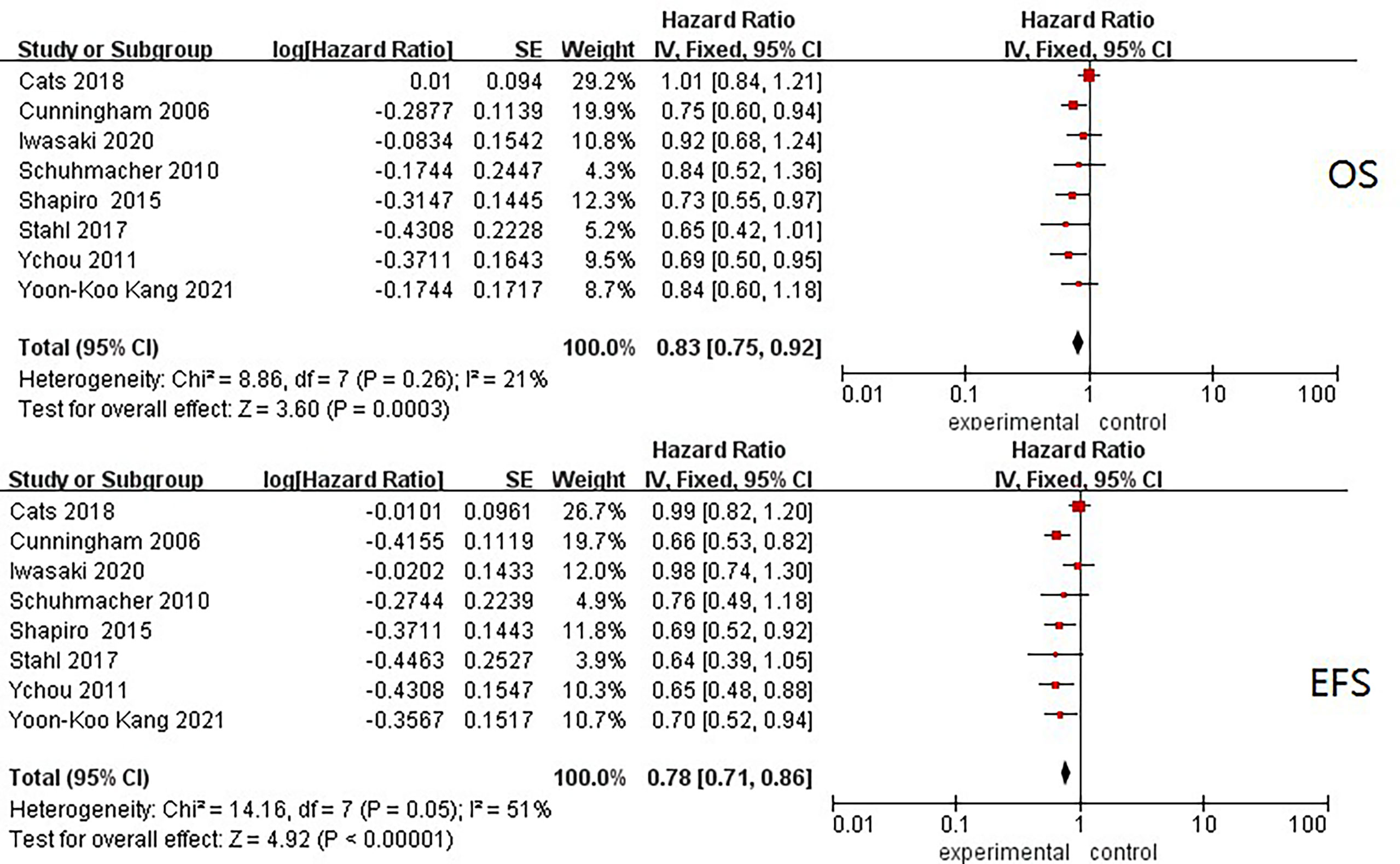
Frontiers | Evaluation of Event-Free Survival Surrogating Overall Survival as the Endpoint in Neoadjuvant Clinical Trials of Gastroesophageal Adenocarcinoma

Statistical considerations and endpoints for clinical lung cancer studies: Can progression free survival (PFS) substitute overall survival (OS) as a valid endpoint in clinical trials for advanced non-small-cell lung cancer? | Semantic

Disease-free and overall survival at 3.5 years for neoadjuvant bevacizumab added to docetaxel followed by fluorouracil, epirubicin and cyclophosphamide, for women with HER2 negative early breast cancer: ARTemis Trial - Annals of

Event-free survival at 24 months is a robust surrogate endpoint for long-term survival in pediatric, adolescent, and adult T cell lymphoblastic lymphoma | SpringerLink

Kaplan Meier plots, showing the difference in overall survival, event... | Download Scientific Diagram

Progression-Free Survival and Overall Survival Beyond 5 Years of NSCLC Patients With Synchronous Oligometastases Treated in a Prospective Phase II Trial (NCT 01282450) - Journal of Thoracic Oncology
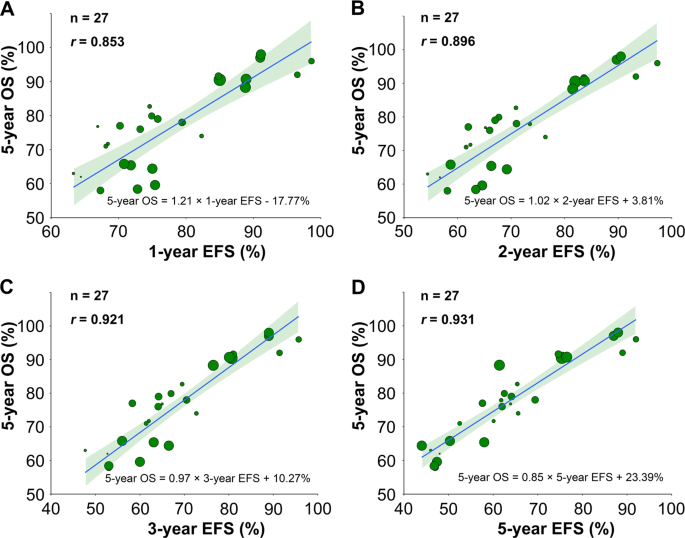
Association of progression-free or event-free survival with overall survival in diffuse large B-cell lymphoma after immunochemotherapy: a systematic review | Leukemia
Long-term follow up of pediatric Philadelphia positive acute lymphoblastic leukemia treated with the EsPhALL2004 study: high white blood cell count at diagnosis is the strongest prognostic factor | Haematologica

Disease-free survival as a surrogate for overall survival in neoadjuvant trials of gastroesophageal adenocarcinoma: Pooled analysis of individual patient data from randomised controlled trials - European Journal of Cancer

Survival and risk factors for mortality in pediatric patients with acute myeloid leukemia in a single reference center in low–middle-income country | SpringerLink

Identification of potential surrogate end points in randomized clinical trials of aggressive and indolent non-Hodgkin's lymphoma: correlation of complete response, time-to-event and overall survival end points - Annals of Oncology

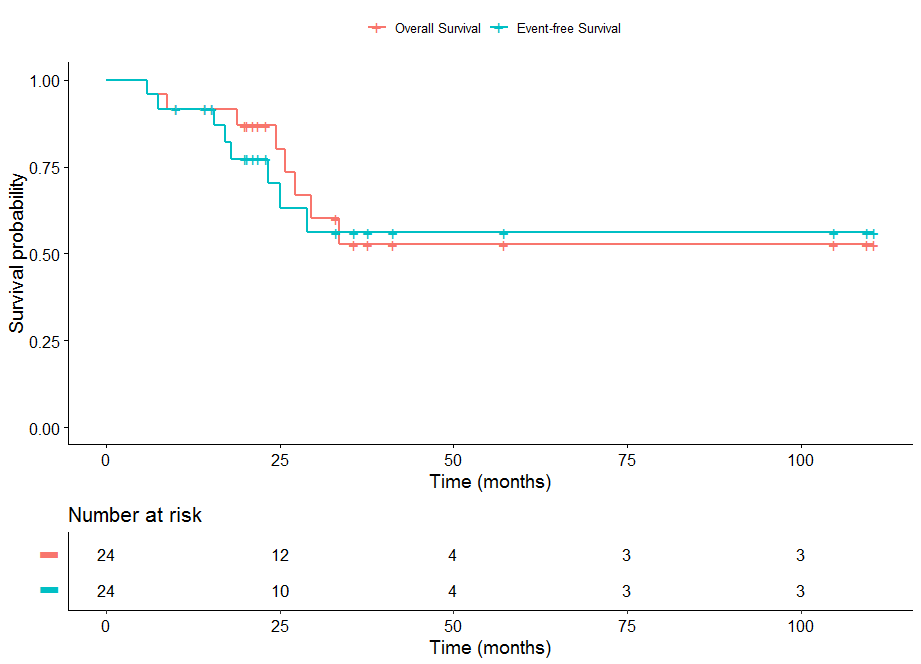

![Fig. 72.1, [Four-year event-free survival (EFS), overall...]. - The EBMT Handbook - NCBI Bookshelf Fig. 72.1, [Four-year event-free survival (EFS), overall...]. - The EBMT Handbook - NCBI Bookshelf](https://www.ncbi.nlm.nih.gov/books/NBK553933/bin/978-3-030-02278-5_72_Fig1_HTML.jpg)

