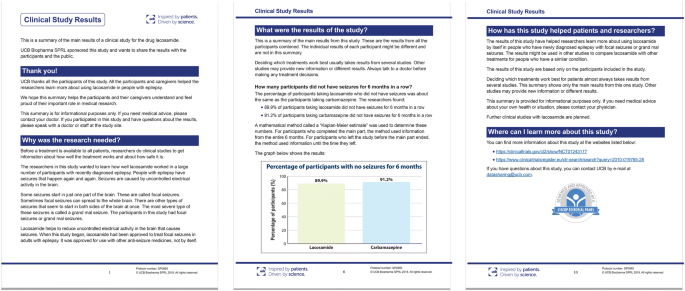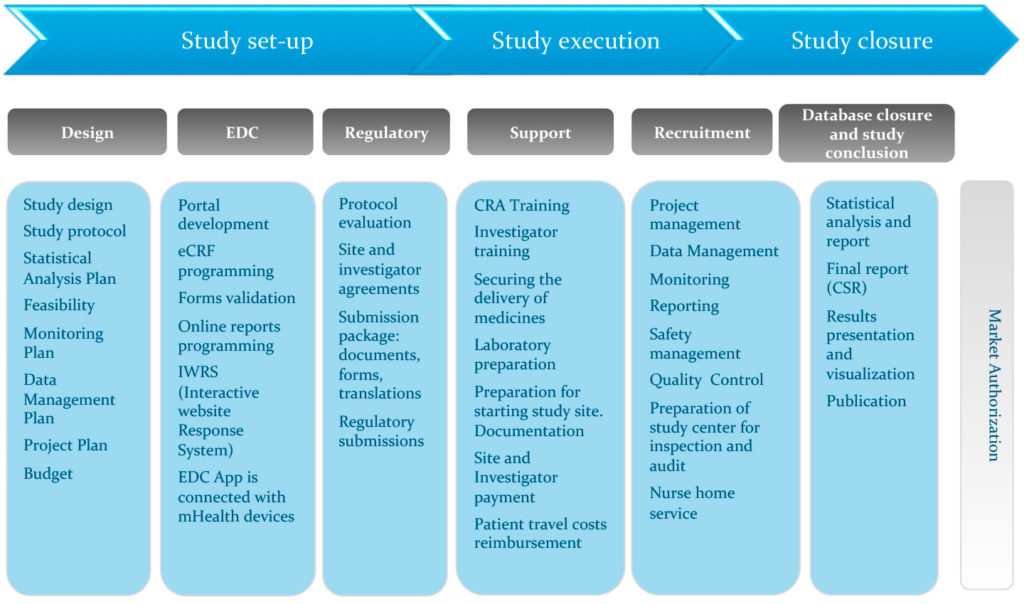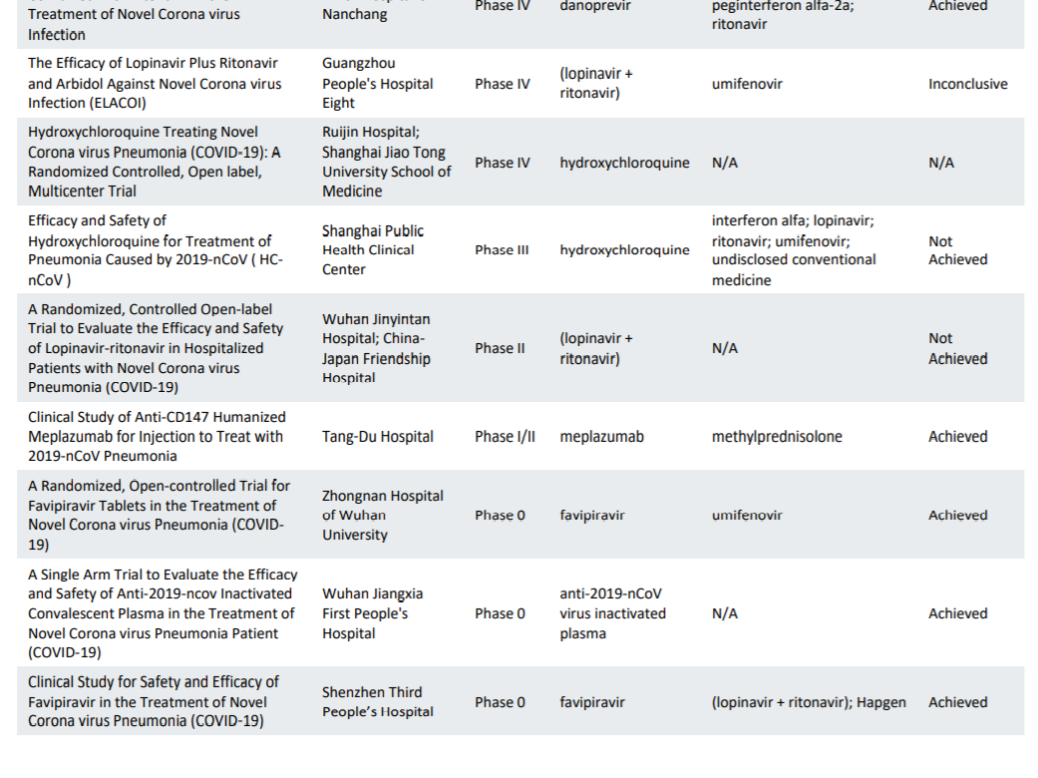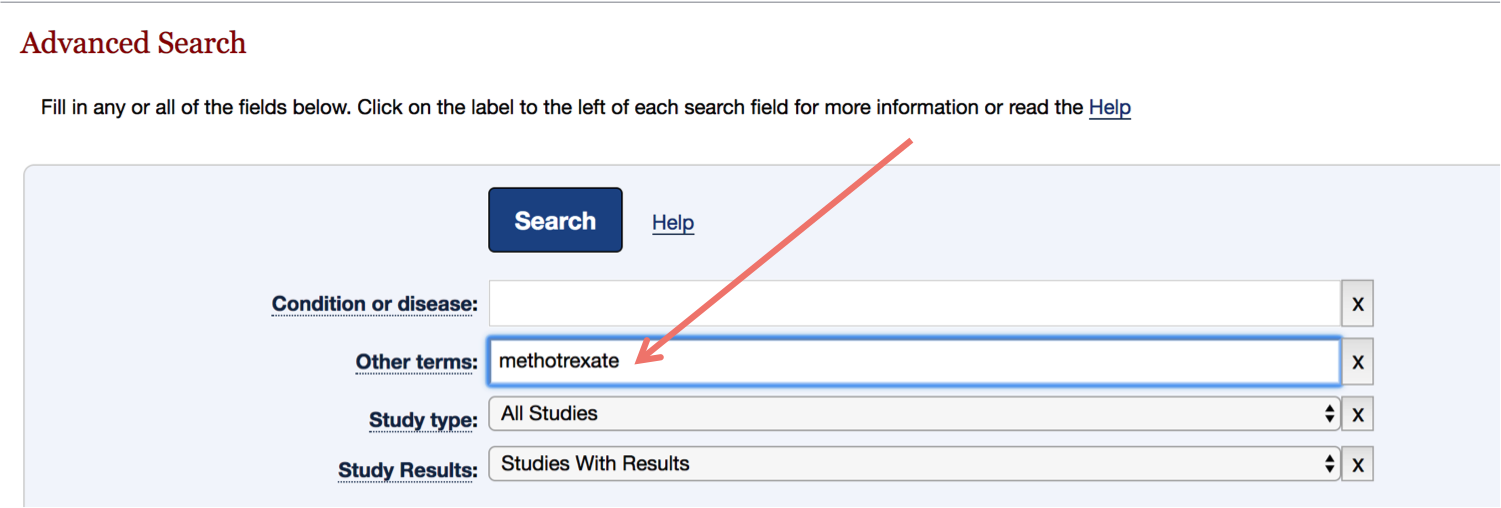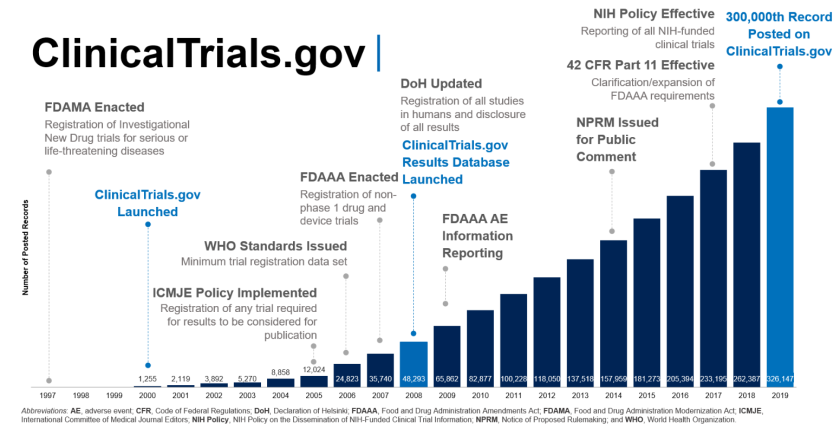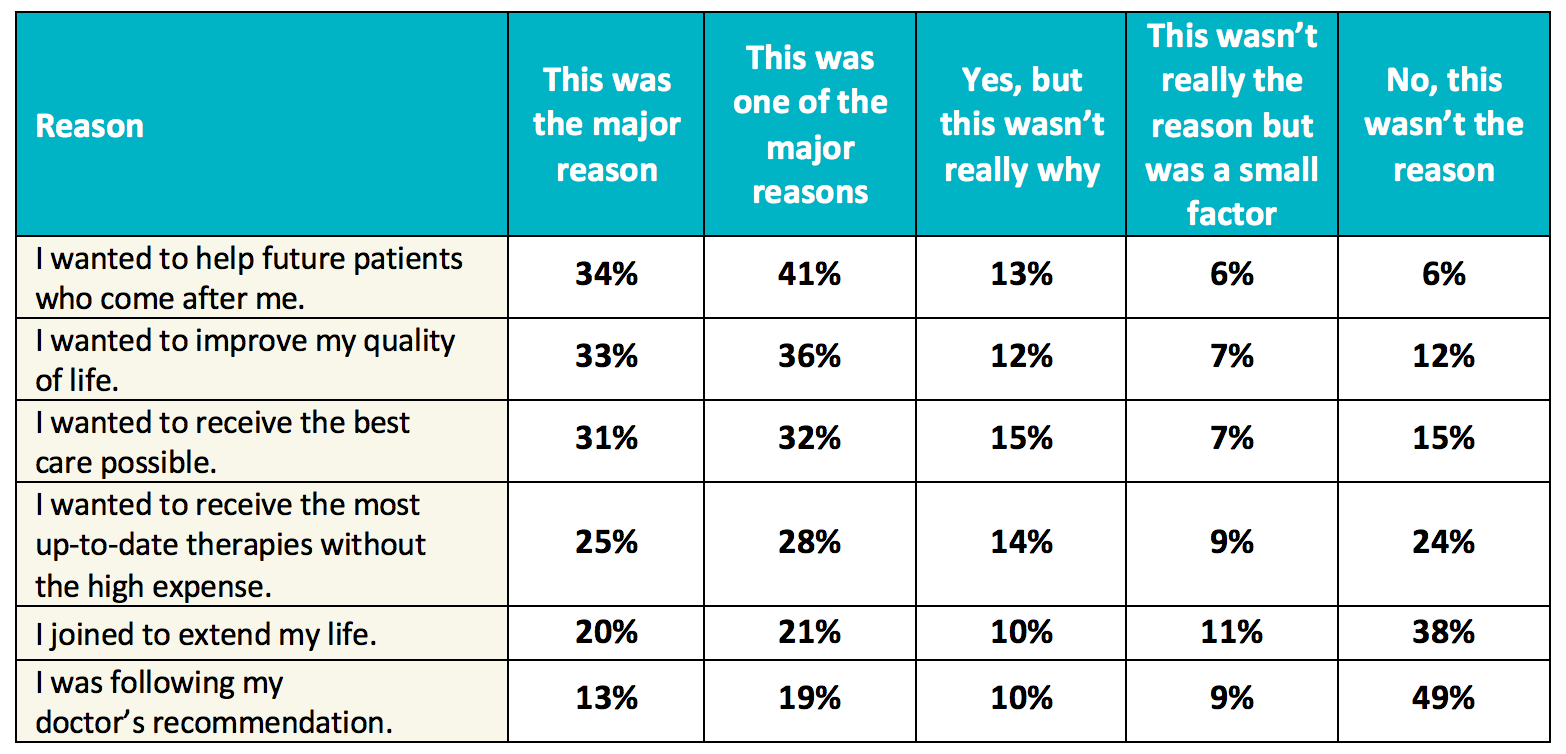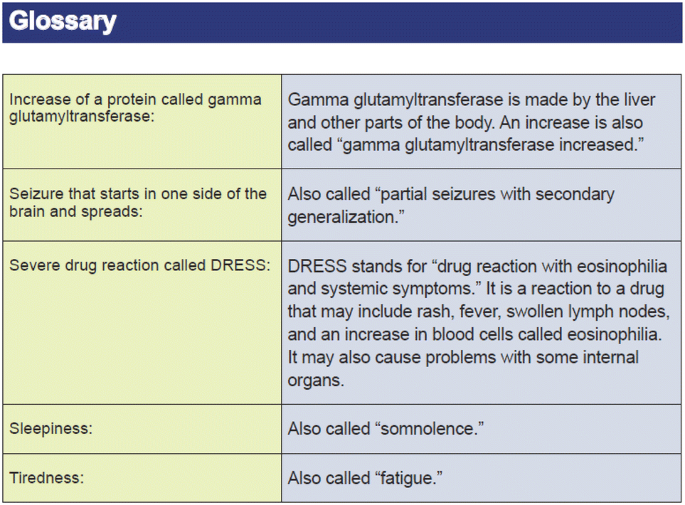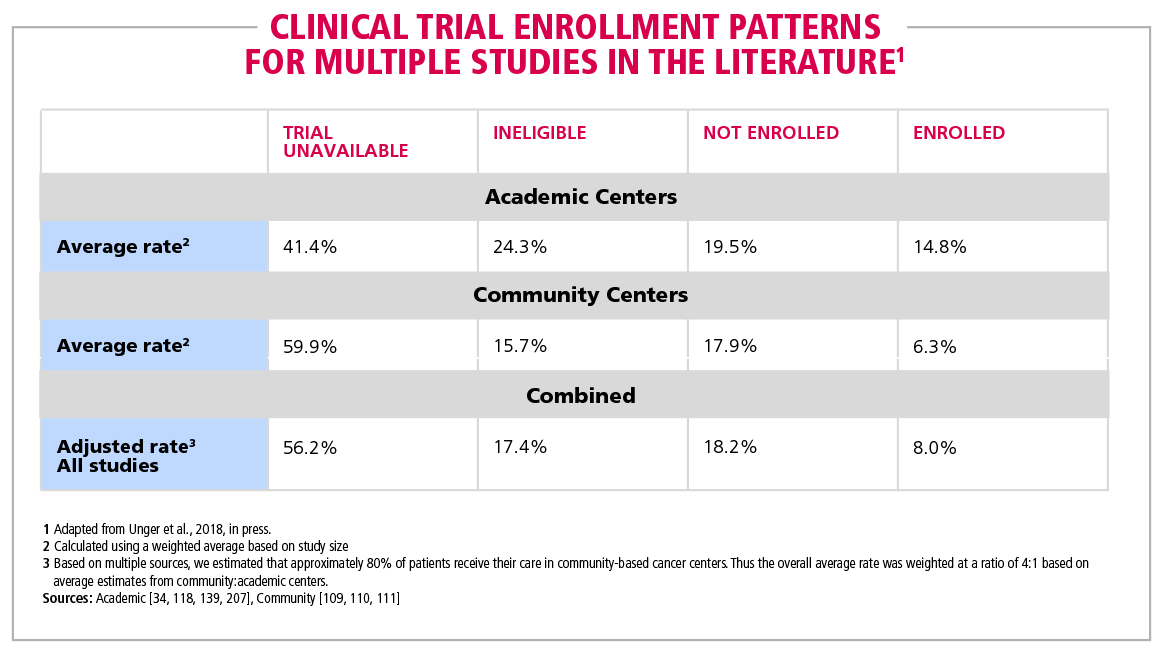
Table 1. Clinical trial enrollment patterns for multiple studies in the literature | American Cancer Society Cancer Action Network
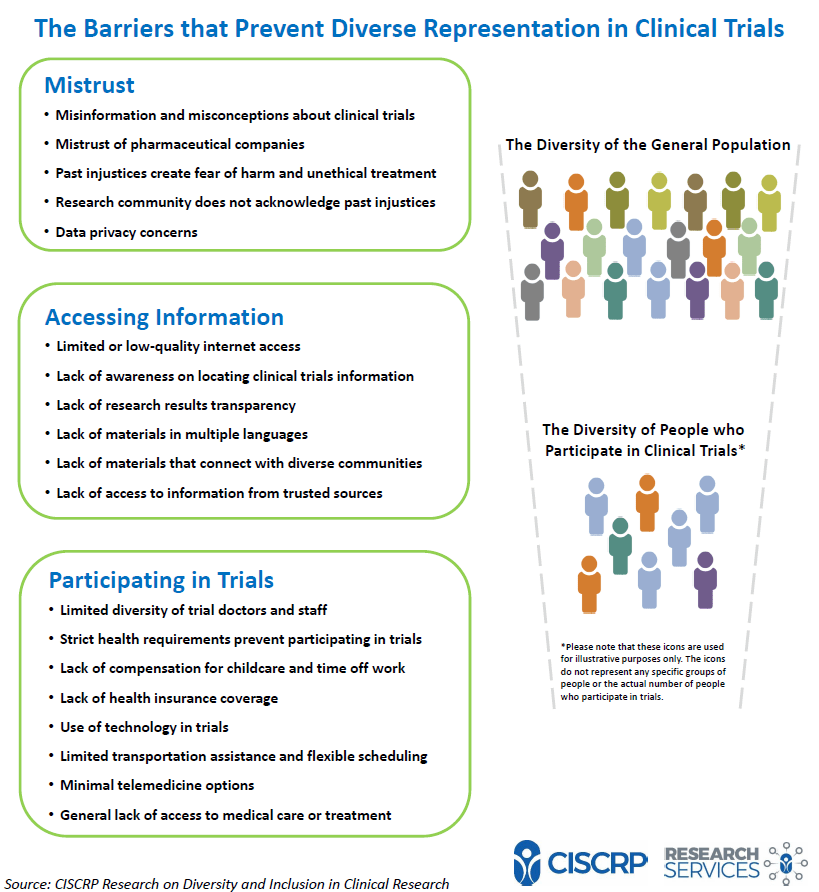
Doing Our Part: Improving Diversity in Clinical Research Participation - Center for Information & Study on Clinical Research Participation

Clinical Trials - Grey Literature in the Health Sciences - Research Guides at University of Alabama - Birmingham
Evidence for the Selective Reporting of Analyses and Discrepancies in Clinical Trials: A Systematic Review of Cohort Studies of Clinical Trials | PLOS Medicine

Statistical controversies in clinical research: limitations of open-label studies assessing antiangiogenic therapies with regard to evaluation of vascular adverse drug events—a meta-analysis - Annals of Oncology