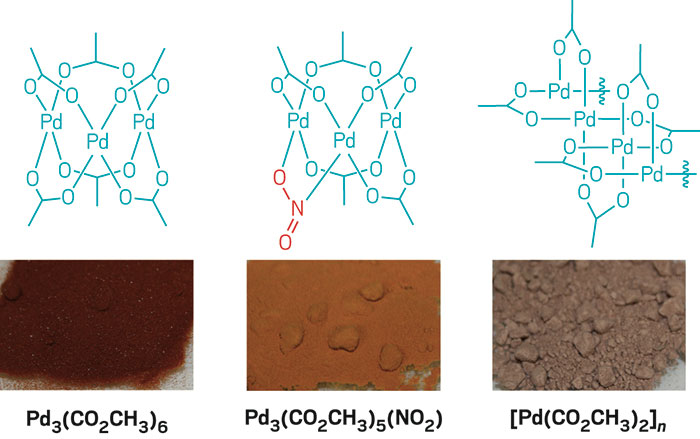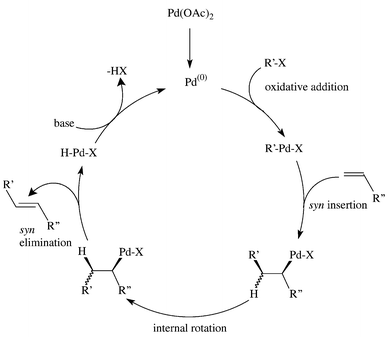
Structural characterisation of solution species implicated in the palladium -catalysed Heck reaction by Pd K-edge X-ray absorption spectroscopy : pall ... - Journal of the Chemical Society, Dalton Transactions (RSC Publishing) DOI:10.1039/B200617K
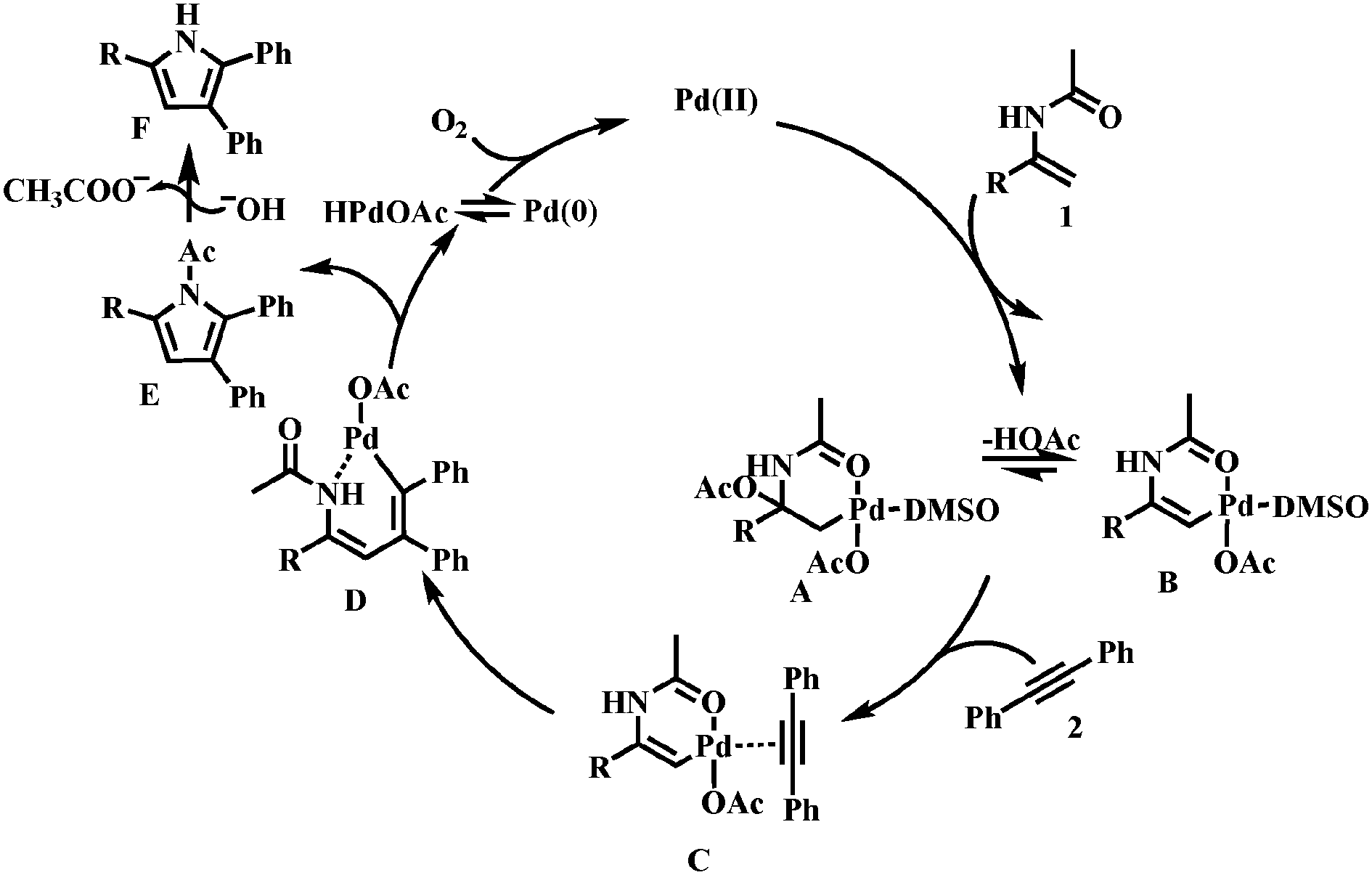
Synthesis of multi-substituted pyrroles using enamides and alkynes catalyzed by Pd(OAc)2 with molecular oxygen as an oxidant - Chemical Communications (RSC Publishing)

Palladium-catalyzed microwave-assisted Hirao reaction utilizing the excess of the diarylphosphine oxide reagent as the P-ligand; a study on the activity and formation of the “PdP2” catalyst in: Pure and Applied Chemistry Volume

Anchoring palladium acetate onto imine-functionalized silica gel through coordinative attachment: An effective recyclable catalyst for the Suzuki–Miyaura reaction in aqueous-isopropanol - ScienceDirect
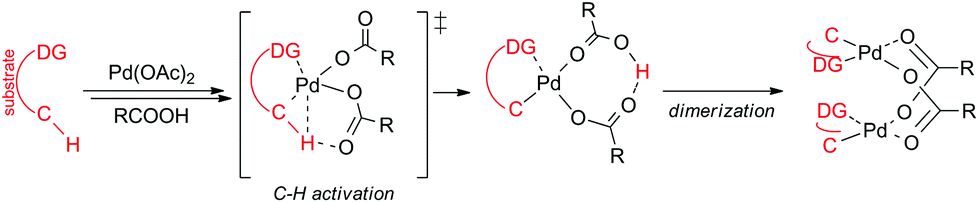
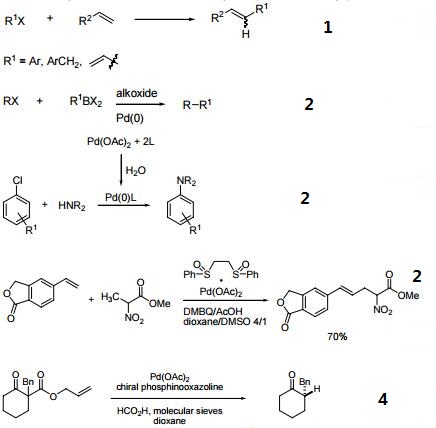
Bi and trinuclear complexes in palladium carboxylate-assisted C–H activation reactions - Dalton Transactions (RSC Publishing) DOI:10.1039/C7DT04269H
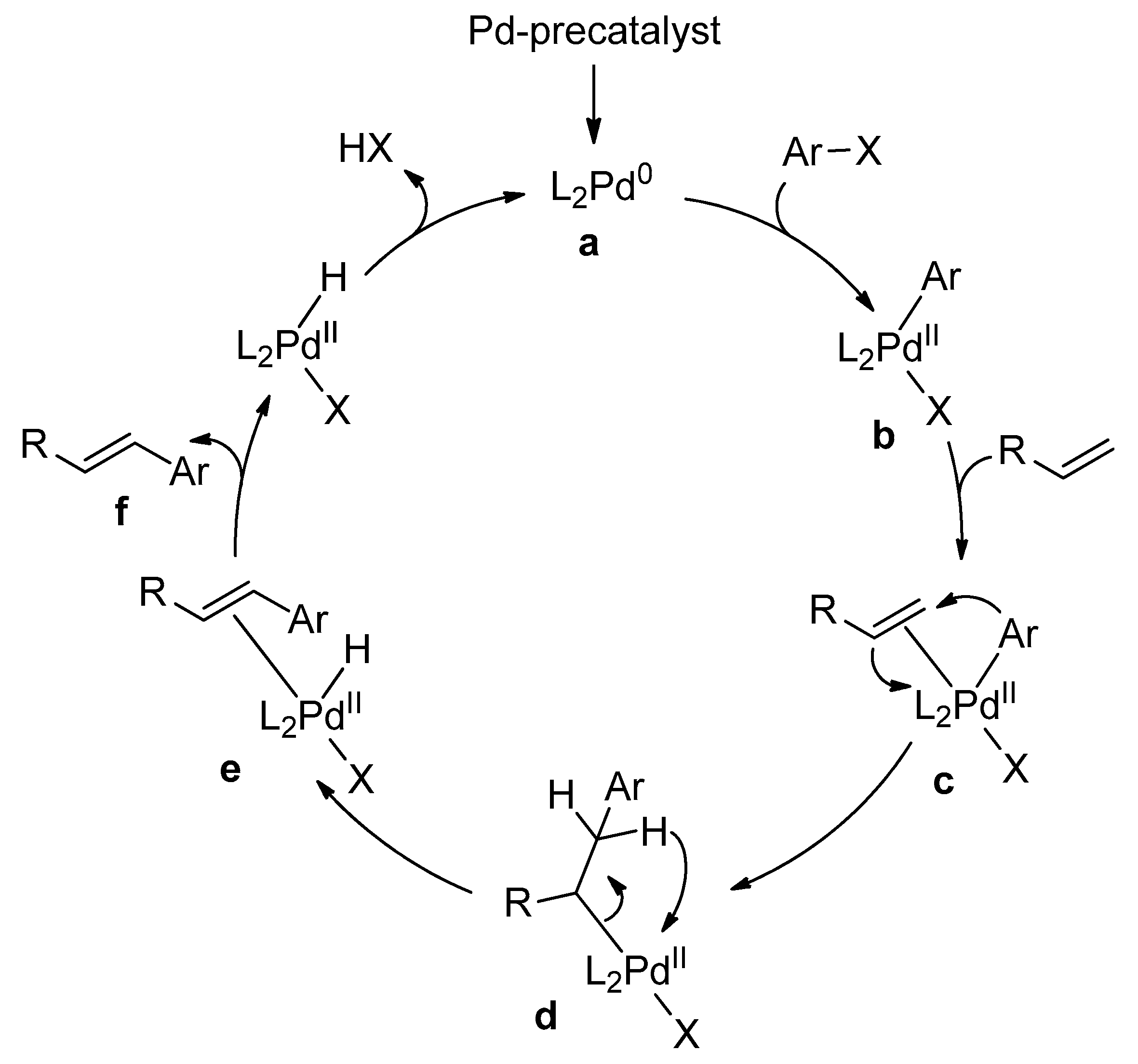
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
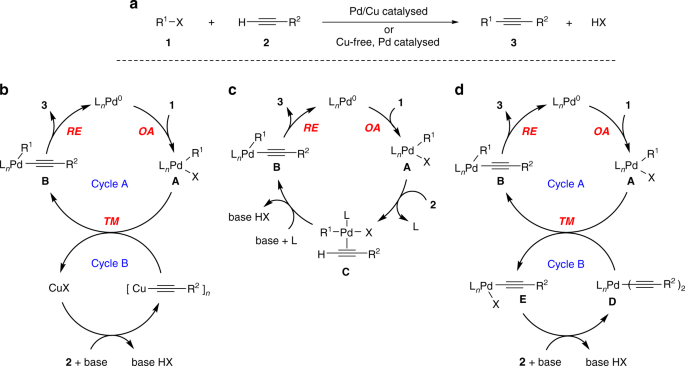
Mechanism of copper-free Sonogashira reaction operates through palladium- palladium transmetallation | Nature Communications

The Heck Reaction and Cinnamic Acid Synthesis by Heterogeneous Catalysis | Johnson Matthey Technology Review

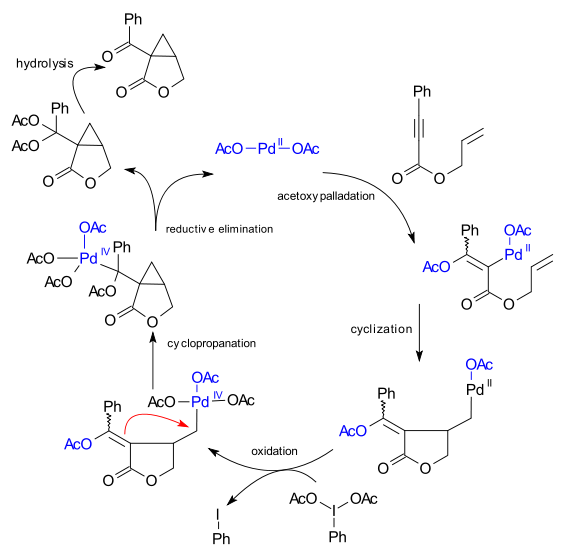
Scheme 4. Proposed mechanism for the palladium(II)-catalyzed oxidative... | Download Scientific Diagram

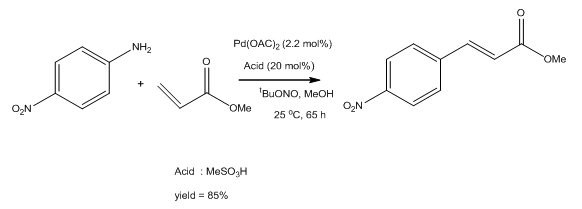
Palladium(II) Acetate Catalyzed Reductive Heck Reaction of Enones; A Practical Approach - Mannathan - 2015 - ChemCatChem - Wiley Online Library