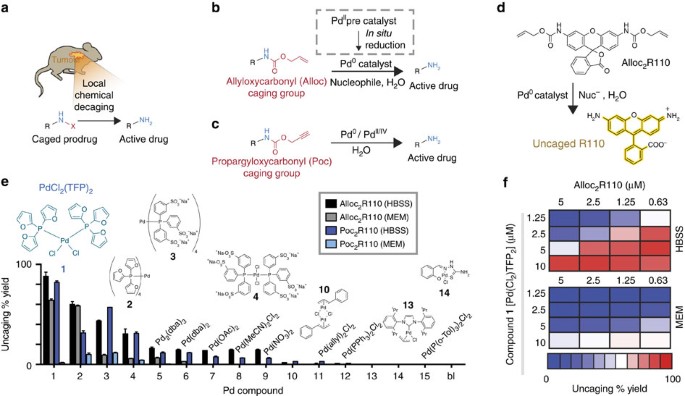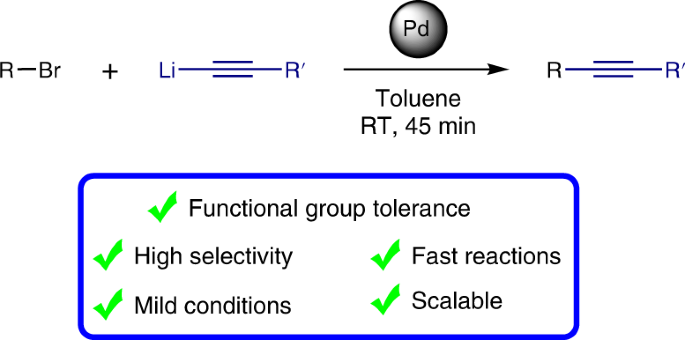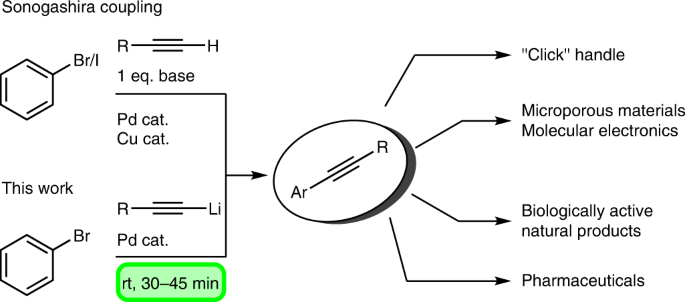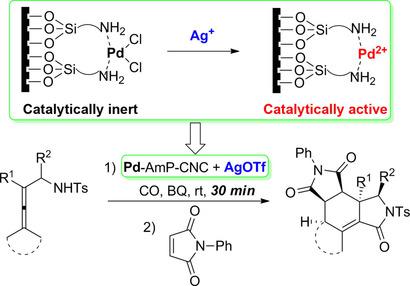
Silver-Triggered Activity of a Heterogeneous Palladium Catalyst in Oxidative Carbonylation Reactions. - Angew. Chem. Int. Ed. - X-MOL

Extracellular Palladium-Catalyzed Dealkylation of 5-Fluoro-1-propargyl-uracil as a Bioorthogonally Activated Prodrug Approach | Request PDF
![PDF] Development and Bioorthogonal Activation of Palladium-Labile Prodrugs of Gemcitabine | Semantic Scholar PDF] Development and Bioorthogonal Activation of Palladium-Labile Prodrugs of Gemcitabine | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7463568d3d77326fd67d807417cdb2dda2f80ba6/3-Figure1-1.png)
PDF] Development and Bioorthogonal Activation of Palladium-Labile Prodrugs of Gemcitabine | Semantic Scholar

PDF) Palladium-Mediated Dealkylation of N-Propargyl-Floxuridine as a Bioorthogonal Oxygen-Independent Prodrug Strategy

Cancer-derived exosomes loaded with ultrathin palladium nanosheets for targeted bioorthogonal catalysis | Nature Catalysis

Development and bioorthogonal activation of palladium-labile prodrugs of gemcitabine. - Abstract - Europe PMC

Cross-coupling reaction on Dha:a)General reaction scheme for the Dha... | Download Scientific Diagram

Cancer-derived exosomes loaded with ultrathin palladium nanosheets for targeted bioorthogonal catalysis. - Abstract - Europe PMC

Illustration of design of photo-responsive bioorthogonal catalysts in... | Download Scientific Diagram
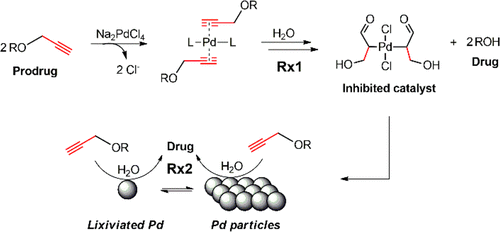
Mechanism of Palladium(II)-Mediated Uncaging Reactions of Propargylic Substrates,ACS Catalysis - X-MOL

Palladium in the Chemical Synthesis and Modification of Proteins - Jbara - 2017 - Angewandte Chemie International Edition - Wiley Online Library

Molecules | Free Full-Text | Palladium-Catalyzed Modification of Unprotected Nucleosides, Nucleotides, and Oligonucleotides | HTML
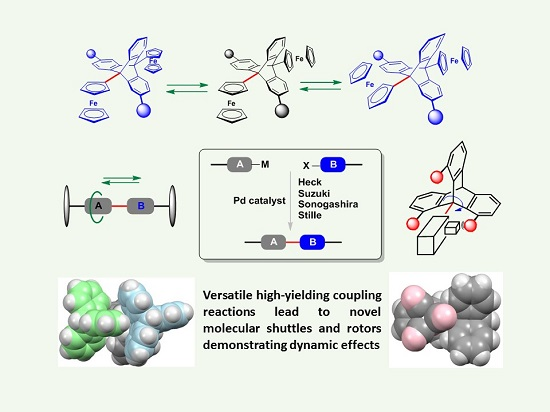
Molecules | Free Full-Text | Palladium-Catalysed Coupling Reactions En Route to Molecular Machines: Sterically Hindered Indenyl and Ferrocenyl Anthracenes and Triptycenes, and Biindenyls | HTML